|
It is helpful to remember that monoatomic ions always have an oxidation number equal to their charge. Though through frequent and repetitive practice, it'll probably happen anyway. You can definitely just have the Periodic Table by your side so that you don't really have to memorize oxidation numbers. In 2011, a study of the Georgia Institute of Technology unveiled the presence of magnesium chlorate on the planet Mars.Arisawaters2D wrote:Is the best way to go about these types of problems memorizing the oxidation numbers for some elements? This means that the chlorate anion exists only as a substitution in the known mineral species, or – eventually – is present in the pore-filling solutions. It should be clearly stated, that currently no chlorate-dominant minerals are known. Further, the evolution of chlorate reduction may be an ancient phenomenon as all perchlorate reducing bacteria described to date also utilize chlorate as a terminal electron acceptor. From a microbial standpoint, the presence of natural chlorate could also explain why there is a variety of microorganisms capable of reducing chlorate to chloride. It is suspected that chlorate and perchlorate may share a common natural formation mechanism and could be a part of the chlorine biogeochemistry cycle. The chlorate was also measured in rainfall samples with the amount of chlorate similar to perchlorate. Natural occurrence Ī recent study has discovered the presence of natural chlorate deposits around the world, with relatively high concentrations found in arid and hyper-arid regions. The heating of the reactants to 50–70 ☌ is performed by the electrical power used for electrolysis. If the electrolysis equipment allows for the mixing of the chlorine and the sodium hydroxide, then the disproportionation reaction described above occurs. The industrial-scale synthesis for sodium chlorate starts from an aqueous sodium chloride solution (brine) rather than chlorine gas. The reaction of cold aqueous metal hydroxides with chlorine produces the chloride and hypochlorite (oxidation number +1) instead. Chlorine, oxidation number 0, forms chloride Cl − (oxidation number −1) and chlorate(V) ClO −ģ (oxidation number +5). In this reaction, chlorine undergoes disproportionation, both reduction and oxidation. Metal chlorates can be prepared by adding chlorine to hot metal hydroxides like KOH: Instead, it is often thought of as a hybrid of multiple resonance structures: The chlorate ion cannot be satisfactorily represented by just one Lewis structure, since all the Cl–O bonds are the same length (1.49 Å in potassium chlorate ), and the chlorine atom is hypervalent. Most pyrotechnic applications that formerly used chlorates now use the more stable perchlorates instead. 
Chlorates were once widely used in pyrotechnics for this reason, though their use has fallen due to their instability. Mixtures of chlorate salts with virtually any combustible material (sugar, sawdust, charcoal, organic solvents, metals, etc.) will readily deflagrate.  
Other oxyanions of chlorine can be named "chlorate" followed by a Roman numeral in parentheses denoting the oxidation state of chlorine: e.g., the ClO −Ĥ ion commonly called perchlorate can also be called chlorate(VII).Īs predicted by valence shell electron pair repulsion theory, chlorate anions have trigonal pyramidal structures.Ĭhlorates are powerful oxidizers and should be kept away from organics or easily oxidized materials. The term can also refer to chemical compounds containing this anion, with chlorates being the salts of chloric acid. 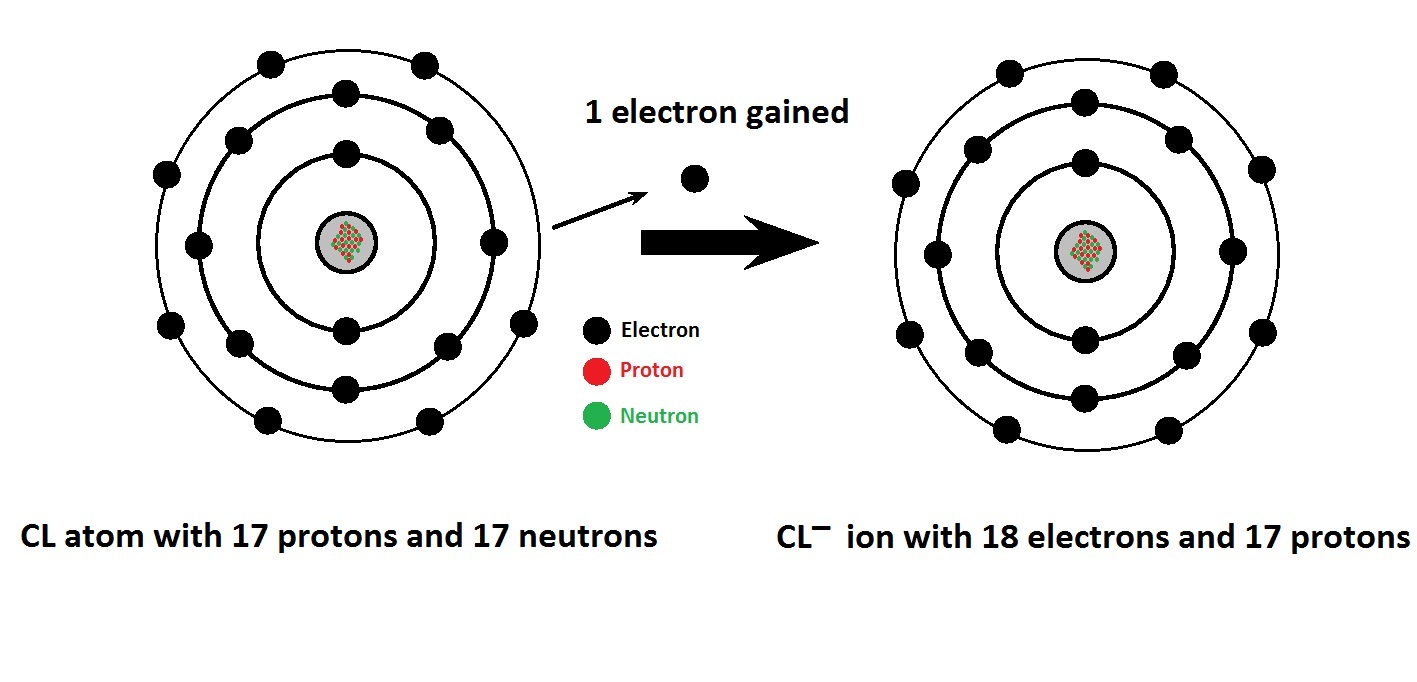
3 anion, whose chlorine atom is in the +5 oxidation state.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
